 Elements in the second row of the periodic table place their electrons in the 2n shell as well as the 1n shell. After the 1 s 1s 1 s 1, s orbital is filled, the second electron shell begins to fill, with electrons going first into the 2 s 2s 2 s 2, s orbital and then into the three p p p p orbitals. The second electron shell, 2n, contains another spherical s s s s orbital plus three dumbbell-shaped p p p p orbitals, each of which can hold two electrons. Hydrogen and helium are the only two elements that have electrons exclusively in the 1 s 1s 1 s 1, s orbital in their neutral, non-charged, state. On the periodic table, hydrogen and helium are the only two elements in the first row, or period, which reflects that they only have electrons in their first shell. This is written out as 1 s 2 1s^ 2 1 s 2 1, s, squared, referring to the two electrons of helium in the 1 s 1s 1 s 1, s orbital. Helium has two electrons, so it can completely fill the 1 s 1s 1 s 1, s orbital with its two electrons. This can be written out in a shorthand form called an electron configuration as 1 s 1 1s^ 1 1 s 1 1, s, start superscript, 1, end superscript, where the superscripted 1 refers to the one electron in the 1 s 1s 1 s 1, s orbital. Hydrogen has just one electron, so it has a single spot in the 1 s 1s 1 s 1, s orbital occupied. The 1 s 1s 1 s 1, s orbital is the closest orbital to the nucleus, and it fills with electrons first, before any other orbital. In other words, even though an element group might break a trend, the elements within the group display periodic properties.The first electron shell, 1n, corresponds to a single 1 s 1s 1 s 1, s orbital. However, the behavior of the noble gases is periodic. The noble gases are an exception to the trend since these elements have filled electron valence shells and electron affinity values approaching zero. Nonmetals usually have higher electron affinities than metals. Electron affinity increases moving across a period and decreases moving down a group. Electron Affinity - This is a measure of readily an atom accepts an electron.Atom and ion sizes shrink moving across a period because the increasing positive charge of the nucleus pulls in the electron shell. 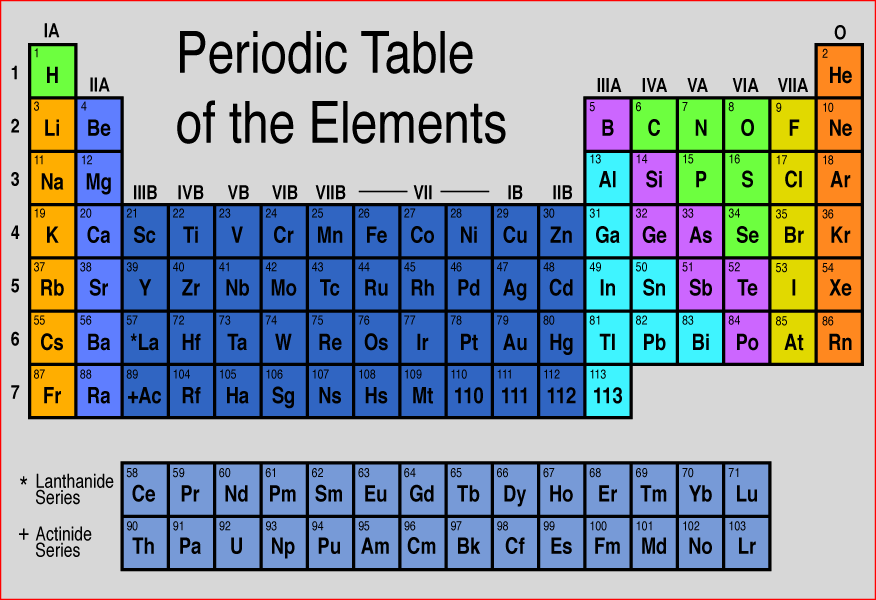
Although it might seem like increasing the number of protons and electrons in an atom would always increase its size, the atom size doesn't increase until a new electron shell is added. Ionic radius is the distance for ions of the atoms and follows the same trend. Atomic radius decreases moving left to right across a period and increases moving down a group.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
